Data dashboard
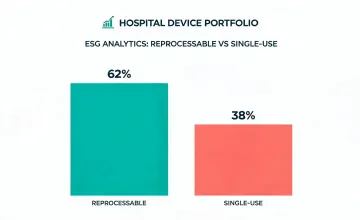
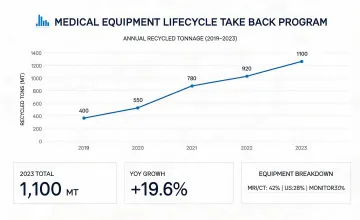
Reporting for commercial medical device programs has to be specific enough for investors, public institutions, and hospital procurement leaders. Ge Healthcare sustainability information is organized around manufacturing impact, device lifecycle, single-use material control, take-back programs, and the documentation trail committees expect during responsible sourcing review.






The download list is structured for sourcing teams that need comparable evidence, not aspirational claims. Each file can be mapped to a supplier questionnaire, value analysis form, or public-sector purchasing review.
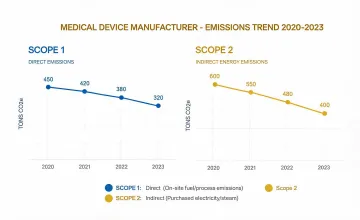
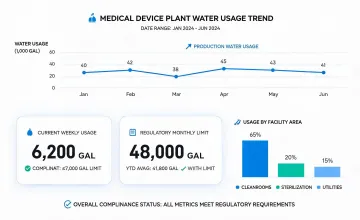
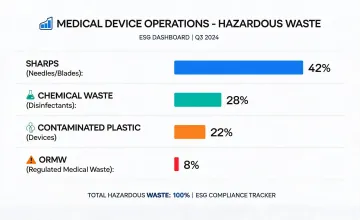
Facility energy, water, waste, and supplier review metrics were standardized for year-over-year comparability.
Climate risk was tied to manufacturing continuity, supplier resilience, and global distribution planning.
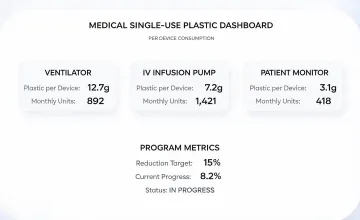
Consumables and procedure pack programs were evaluated for packaging reduction, shelf-life protection, and responsible waste handling.
Product safety, lifecycle, access, and environmental metrics were aligned with medical equipment reporting categories.
Hospital sourcing teams can request the ESG packet alongside service, cybersecurity, and clinical performance files.
Use the contact form to request the sourcing packet for patient monitoring, home care, rehabilitation, and infection-control product groups.
Request ESG Packet